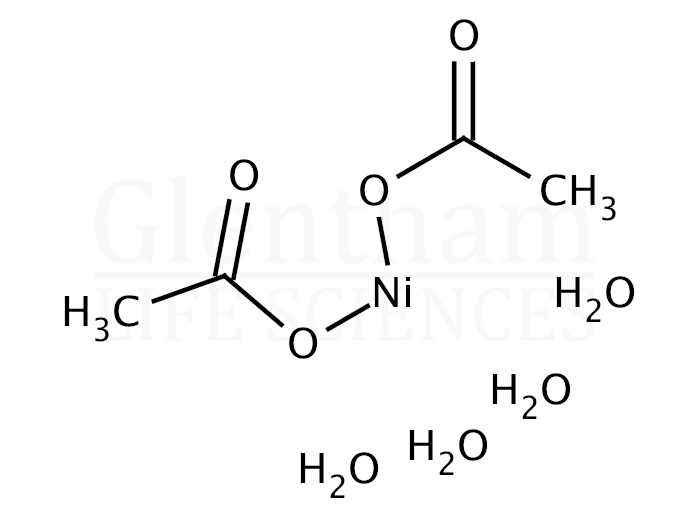
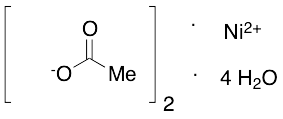
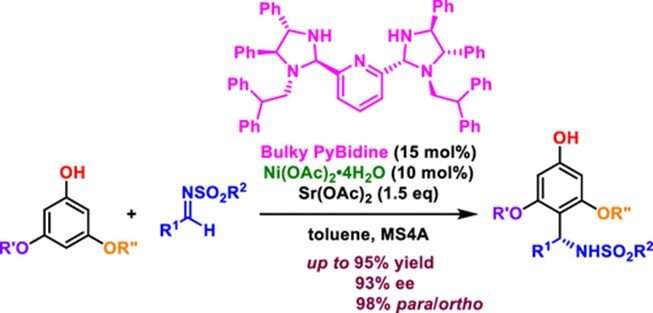
Synthesis, crystal structure, spectroscopic investigations, and computational studies of Ni(II) and Pd(II) complexes with asymmetric tetradentate NOON Schiff base ligand | SpringerLink

Ni‐Catalyzed Regioselective C‐5 Halogenation of 8‐Aminoquinoline and Co‐Catalyzed Chelation Assisted C−H Iodination of Aromatic Sulfonamides with Molecular Iodine - Fernandes - 2022 - Chemistry – An Asian Journal - Wiley Online Library

Stereospecific/stereoselective nickel catalyzed reductive cross-coupling: An efficient tool for the synthesis of biological active targeted molecules - ScienceDirect

SYNTHESIS, SPECTROSCOPIC INVESTIGATION AND CATALYTIC STUDIES OF NICKEL(II) AROMATIC AZOMETHINE COMPLEXES | Semantic Scholar
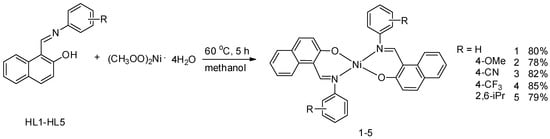
Polymers | Free Full-Text | Mononuclear Nickel(II) Complexes with Schiff Base Ligands: Synthesis, Characterization, and Catalytic Activity in Norbornene Polymerization
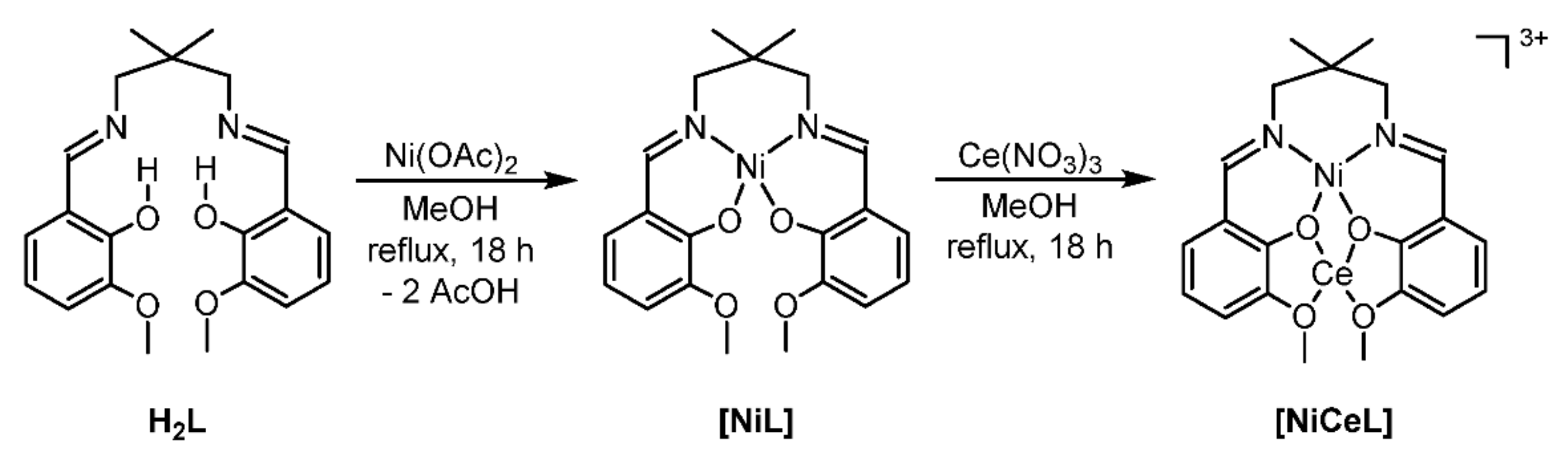
IJMS | Free Full-Text | Mild and Efficient Heterogeneous Hydrogenation of Nitroarenes Facilitated by a Pyrolytically Activated Dinuclear Ni(II)-Ce(III) Diimine Complex
![Naked d-orbital in a centrochiral Ni(II) complex as a catalyst for asymmetric [3+2] cycloaddition | Nature Communications Naked d-orbital in a centrochiral Ni(II) complex as a catalyst for asymmetric [3+2] cycloaddition | Nature Communications](https://media.springernature.com/m685/springer-static/image/art%3A10.1038%2Fncomms14875/MediaObjects/41467_2017_Article_BFncomms14875_Fig5_HTML.jpg)
Naked d-orbital in a centrochiral Ni(II) complex as a catalyst for asymmetric [3+2] cycloaddition | Nature Communications

One to Find Them All: A General Route to Ni(I)–Phenolate Species | Journal of the American Chemical Society

Nickel hydroxides and related materials: a review of their structures, synthesis and properties | Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences

Carboxylation of the Ni–Me Bond in an Electron-Rich Unsymmetrical PCN Pincer Nickel Complex | Organometallics

General Method for the Amination of Aryl Halides with Primary and Secondary Alkyl Amines via Nickel Photocatalysis

Catalysts | Free Full-Text | Synthesis and Characterization of Nickel(II) Homogeneous and Supported Complexes for the Hydrogenation of Furfural to Furfuryl Alcohol

Reactions of Schiff Base‐Substituted Diselenides and ‐tellurides with Ni(II), Pd(II) and Pt(II) Phosphine Complexes - Roca Jungfer - 2020 - European Journal of Inorganic Chemistry - Wiley Online Library


![Cations of (Top) [Ni 2 L1(µ-OAc) 2 ] + (Moffat et al., 2014), and... | Download Scientific Diagram Cations of (Top) [Ni 2 L1(µ-OAc) 2 ] + (Moffat et al., 2014), and... | Download Scientific Diagram](https://www.researchgate.net/publication/327874910/figure/fig2/AS:674929695539200@1537927187016/Cations-of-Top-Ni-2-L1-OAc-2-Moffat-et-al-2014-and-Bottom-Ni-2-Me-4.png)